1. Introduction
The advancement of medicine and the emergence of new viruses, such as SARS-CoV-2, have led to significant progress in vaccine development.[1] A recent study revealed that millions of doses of the COVID-19 vaccine have been administered globally, achieving herd immunity and playing crucial roles in public health interventions.[2, 3] The use of vaccines have mild adverse events, including fever, fatigue, nausea, and myalgias, with numerous reports of more serious health issues.[4] Biological sex differences influence vaccine responses, indicating the need for sex-specific administration, especially with reports of sex-specific adverse events related to COVID-19 vaccines.[5, 6]
There was a reported increase in the length of menstrual cycles following COVID-19 vaccination.[7] In addition, human papillomaviruses (HPVs) have also been linked to semen infections, which contribute to male infertility.[8] Considering these adverse events, there is still a lack of comparative analysis on sex-specific reproductive disorders that occur as adverse events of vaccines.[8] Understanding these associations is crucial, as reproductive disorders are increasing and negatively impact all aspects of quality of life, including physical health and social functioning, as well as conception.[9, 10] This research gap is particularly important given that males and females have distinct immune responses to vaccinations, influenced by biological and hormonal differences.[11]
Therefore, we aimed to investigate the vaccines that most commonly influence sex-specific reproductive system disorders, focusing on menstrual disorders in females and sexual dysfunction in males.[12] Our study also intended to improve vaccine efficacy and safety for both sexes and propose differentiated vaccination strategies that consider sex-specific reproductive disorders.
2. METHODS
The use of this data in confidence was approved by Kyung Hee University. As this study was based on publicly available data, ethics approval and consent were not required.
Data on vaccine-associated menstrual disorders and male sexual dysfunction were collected, encompassing 19 vaccine categories, including: (1) anthrax vaccines; (2) cholera vaccines; (3) diphtheria, tetanus toxoids, pertussis, polio, and Hemophilus influenza type b (DTaP-IPV-Hib) vaccines; (4) meningococcal vaccines; (5) pneumococcal vaccines; (6) tuberculosis vaccines; (7) typhoid vaccines; (8) encephalitis vaccines; (9) influenza vaccines; (10) hepatitis A vaccines; (11) hepatitis B vaccines; (12) measles, mumps, and rubella (MMR) vaccines; (13) rotavirus diarrhea vaccines; (14) zoster vaccines; (15) papillomavirus vaccines; (16) COVID-19 mRNA vaccines; (17) Ad5-vectored COVID-19 vaccines; (18) inactivated whole-virus COVID-19 vaccines; and (19) others (brucellosis, dengue vaccines, Ebola, leptospirosis, plague, rabies, smallpox, tuberculosis, typhus, and yellow fever vaccines). Only vaccines classified as ‘suspected’ in compliance with WHO guidelines for causality assessment were considered for analysis of disproportionate associations with menstrual disorders and male sexual dysfunction. Analysis was conducted using the Medical Dictionary for Regulatory Activities 26.0.
Our study investigated cases of suspected vaccine-associated menstrual disorders and male sexual dysfunction. We relied primarily on personal safety reports collected from various sources, including patients, healthcare professionals, and pharmaceutical companies, during the post-marketing period.
The dataset contains patient demographics such as age (0-11, 12-17, 18-44, 45-64, ≥65 years, and unknown) and sex, as well as administrative information including reporting regions, reporting years, and reporter qualifications (health professionals, non-health professionals, and unknown). The dataset also includes comprehensive information on vaccines, including vaccine type, suspected vaccine, and details of adverse drug reaction (i.e., time to onset [TTO] of reaction and fatal outcomes [recovered/recovering, fatal, and unknown]).
To explore potential associations between various vaccines in the data set and reports of menstrual disorders and male sexual dysfunction, an examination was conducted. The dataset from 1969 to 2023 was categorized into two groups (case and non-case), and disproportionality analysis was performed on each vaccine.[13] This analysis compares the proportion of adverse events reported for a specific vaccine to that of a control group, which consists of all vaccines in the database. The comparison utilizes the total number of AEs for each vaccine as the denominator to identify any significant associations between the vaccine and AEs.[14] To investigate which vaccines were significantly associated with menstrual disorder and male sexual dysfunction, two standard measures of disproportionate analysis were employed: information components (IC) and reporting odds ratio (ROR).[15] The IC was calculated for case-non-case analysis using a Bayesian confidence propagation neural network. The anticipated quantity of reports and the observed occurrences for every vaccine-adverse event combination were considered in the computation process, where IC0.25 signifies the lower limit of the 95% confidence interval (CI). Positive IC0.25 values (i.e., IC0.25 > 0) denote statistical significance.[16] The disproportionality association was further assessed through the ROR, a metric based on the number of AEs and the vaccine’s contingency table. The ROR was derived by comparing the frequency of specific AEs for a targeted vaccine against the likelihood of the same AEs occurring with all other vaccines in the database. If the lower bound of the ROR’s 95% CI exceeded 1, a significant association between the vaccine and a particular AE was established. The study also employed the unpaired Kruskal-Wallis test for continuous variables and Fisher’s exact test for categorical variables to compare the vaccine with all reported cases. Statistical significance was considered when the two-sided p-value was less than 0.05. All analyses utilized SAS (version 9.4; SAS Inc., Cary, NC, USA), ensuring methodological rigor.
3. Results
A total of 4,939,712 female participants were reported in the full database, with 147,158 of them having vaccine-associated menstrual disorders as reported. There was a significant increase in vaccine-associated menstrual disorders reported from 2020 to 2023 (Table 1). The world was categorized into six different geographical locations. The European region accounted for 77.76% of all vaccine-associated menstrual disorders, followed by the Americas (19.22%), Western Pacific (2.80%), African (0.15%), Eastern Mediterranean (0.05%), and Southeast Asia regions (0.02%). COVID-19 mRNA vaccines had the most significant impact on menstrual disorders (87.67%), followed by Ad5-vectored COVID-19 vaccines (10.42%). Papillomavirus vaccines was responsible for 1.41% of vaccine-associated menstrual disorders, while anthrax vaccines accounted for 0.01%. The prognosis was good for most individuals, with only two reported cases of fatal outcomes (Table 1).
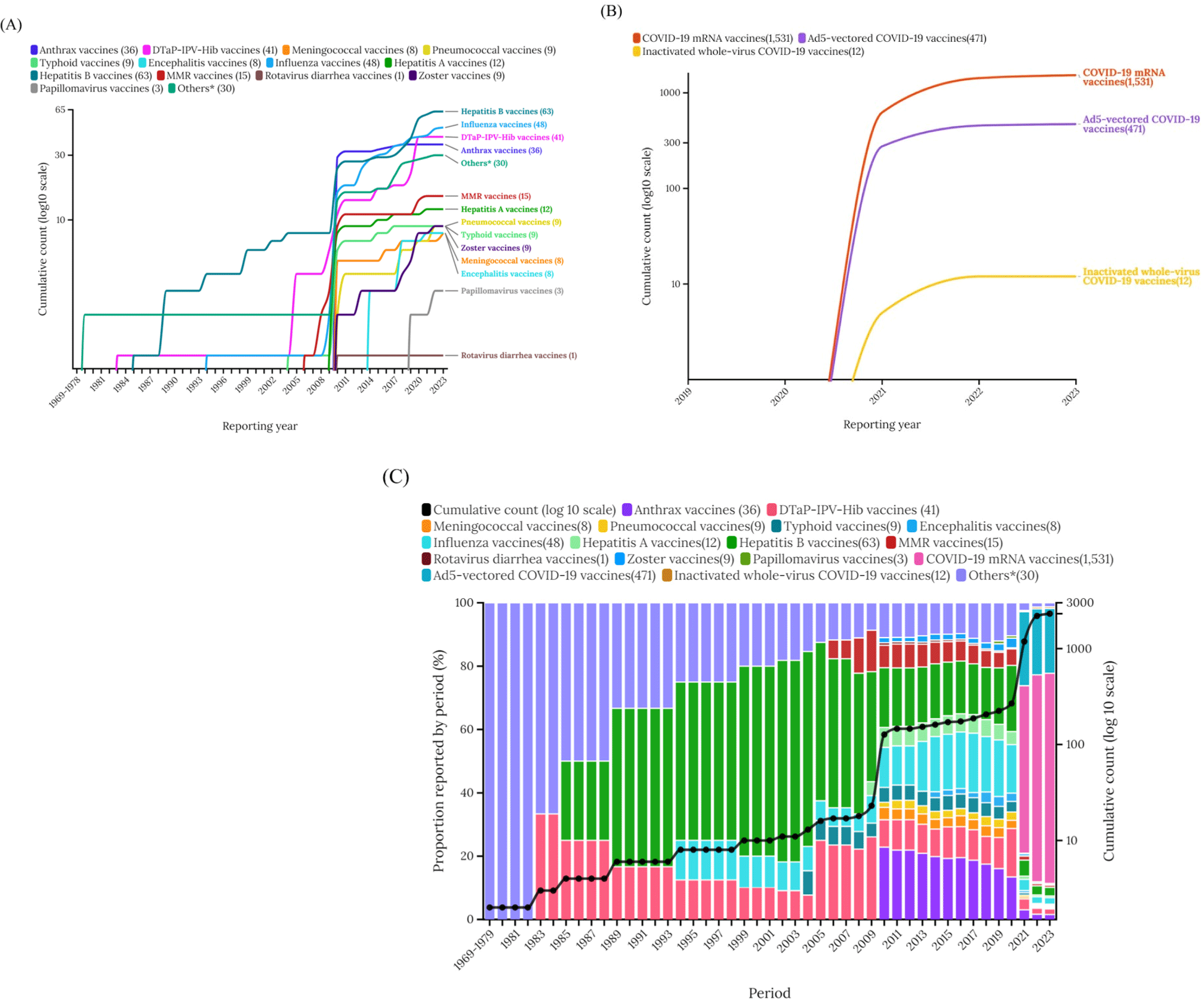
For males, a total of 2,964,055 individuals were reported to the database. Of these, 2,306 individuals reported male sexual dysfunction. Notably, there was a sharp increase in reports of male sexual dysfunction from 2020 to 2023 compared to the period from 1969 to 2019 (Table 1). The world was categorized into six different geographical locations. The European Region accounted for 44.02% of all vaccine-associated male sexual dysfunction disorders, followed by the Americas (43.71%), African (8.41%), Western Pacific (2.60%), Southeast Asia (0.74%) and Eastern Mediterranean (0.52%) regions. COVID-19 mRNA vaccines had the most significant impact on male sexual dysfunction disorders (66.39%), followed by Ad5-vectored COVID-19 vaccines (20.42%). Anthrax vaccines influenced 1.56%. The prognosis was good for most individuals, with zero reported cases of fatal outcomes (Table 1).
Menstrual disorders showed a strong association with specific vaccines. COVID-19 mRNA vaccines had the highest association (ROR [95% CI], 49.51 [48.91-50.13]; IC [IC0.25], 3.36 [3.35]), followed by papillomavirus vaccines (ROR, 6.32 [6.05-6.60]; IC, 2.62 [2.55]), Ad5-vectored COVID-19 vaccines (ROR, 4.44 [4.36-4.51]; IC, 2.02 [1.99]), and anthrax vaccines (ROR, 2.82 [1.85-4.29]; IC: 1.43 [0.72]). Based on age, the papillomavirus vaccine, COVID-19 mRNA vaccine, and Ad5-vectored COVID-19 vaccine showed the highest vaccine-associated menstrual disorder symptoms in the 0-11-year-old group (IC [IC0.25], 4.54[4.15]; 5.46[5.11]; 3.28[1.87], respectively). Additionally, the 45-64-year-old group also exhibited significant associations with these vaccines and menstrual disorder symptoms (4.30[4.28]; 3.28[2.26]; 3.27[3.21]) (Table 2). Male sexual dysfunction was also associated with certain vaccines. The anthrax vaccine exhibited the strongest association (ROR: 4.22 [3.04-5.85]; IC: 2.01[1.45]), followed by COVID-19 mRNA vaccines (ROR: 2.30 [2.18-2.42]; IC: 1.08[1.00, and Ad5-vectored COVID-19 vaccines (ROR: 1.98 [1.81-2.17]; IC: 0.95[0.80]) (Table 3).
Abbreviations: CI, confidence interval; DTaP-IPV-Hib, diphtheria, tetanus toxoids, pertussis, polio, and Hemophilus influenza type b; IC, information component; MMR, measles, mumps, and rubella; ROR, reported odds ratio.
Vaccine associated menstrual disorders saw a sharp increase in 2010 and again in 2020. Before 2010, there was little association between vaccines and menstrual disorders, but the rise in such cases after 2010 was attributed to the widespread administration of papillomavirus vaccines. From 2020 onwards, the introduction of COVID-19 mRNA vaccines and Ad5-vectored COVID-19 vaccines showed a high association with reports of menstrual disorders (Fig. 1). Similarly, there was a sharp increase in vaccine-associated male sexual dysfunction after 2020, attributed to the introduction of COVID-19 mRNA vaccines and Ad5-vectored COVID-19 vaccines (Fig. 2).
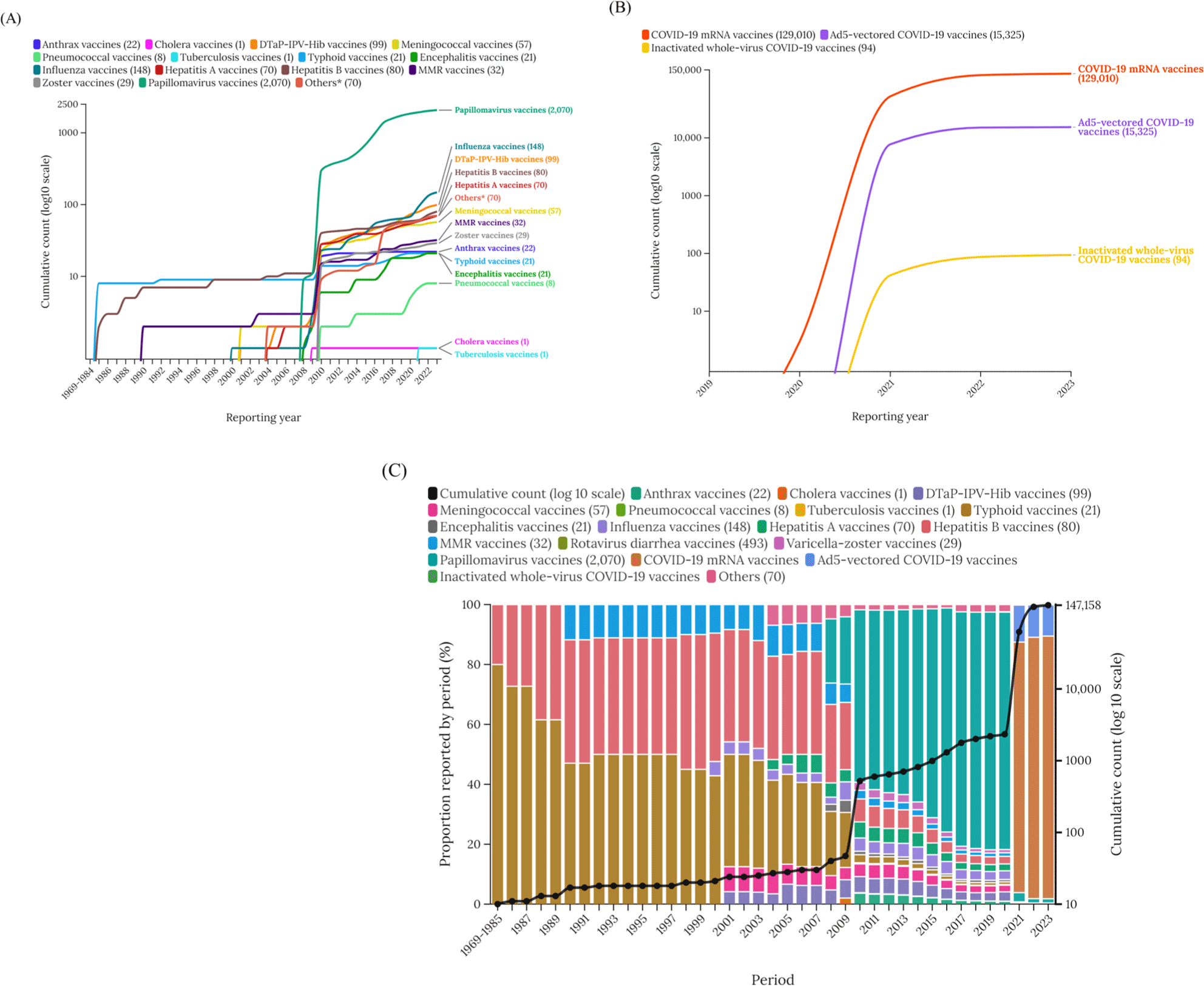
Vaccine-associated menstrual disorder and male sexual dysfunction are classified as endocrine disease. Regarding endocrine adverse effects, the following rates were observed in females: COVID-19 mRNA vaccines at 0.4, papillomavirus vaccines at 3.5, Ad5-vectored COVID-19 at 0.3, and anthrax vaccines at 4.5. These findings suggested that endocrine-related conditions show a certain level of adverse effects with papillomavirus vaccines and anthrax vaccines. In terms of endocrine complications, the following rates were observed in males: COVID-19 mRNA vaccines at 1.9, papillomavirus vaccines at 0, Ad5-vectored COVID-19 at 1.3, and anthrax vaccines at 30.6. These findings suggested that endocrine-related conditions show a certain level of adverse effects with COVID-19 mRNA vaccines, papillomavirus vaccines, Ad5-vectored COVID-19 vaccines, anthrax vaccines.
4. Discussion
Our study examined the impact of various vaccines globally on menstrual disorders and male sexual dysfunction. This study is the first large-scale, longitudinal research that investigated these specific effects. Vaccine-associated menstrual disorders have significantly increased since 2010, coinciding with the widespread administration of papillomavirus vaccines. Furthermore, this trend intensified after 2020 with the introduction of COVID-19 mRNA vaccines and Ad5-vectored COVID-19 vaccines. Similarly, male sexual dysfunction has increased after the introduction of the anthrax vaccine in 2010 and further increased significantly following the introduction of COVID-19 mRNA vaccines and Ad5-vectored COVID-19 vaccines in 2020. While vaccines can lead to an increase in menstrual disorders and male sexual dysfunction, the fatality rate associated with these adverse effects is extremely low, so there is no need to be concerned about getting vaccinated. Our findings suggest that menstrual disorders were most significantly influenced by COVID-19 mRNA vaccines, followed by associations with papillomavirus vaccines, Ad5-vectored COVID-19 vaccines and anthrax vaccines. Among the vaccines identified, the risk of menstrual disorders was particularly higher in the 0-11 and 45-64-year-old age groups. In the case of male sexual dysfunction, it has a significant association with specific vaccines, with the anthrax vaccine having the highest magnitude, followed by the COVID-19 mRNA vaccines and Ad5-vectored COVID-19 vaccines.
Our study suggests that menstrual disorders are associated with COVID-19 mRNA vaccines, papillomavirus vaccines, Ad5-vectored COVID-19 vaccines, and anthrax vaccines. This can be explained by various hypotheses. The systemic immune response triggered by COVID-19 vaccination might disrupt various pathways involved in the menstrual cycle.[7] Menstruation is regulated by hormones, inflammation, and interactions between tissues and cells. Vaccination-induced systemic inflammatory reactions could potentially interfere with these processes, leading to menstrual irregularities or breakthrough bleeding due to hormonal changes. Such interference might influence the hypothalamic–pituitary–ovarian axis, temporarily altering menstrual patterns. Similar disruptions have been observed following natural SARS-CoV-2 infections, suggesting that stress from acute infection or the virus itself could suppress ovarian function or directly impact it.[7]
Associations with Papillomavirus vaccines have also been observed, with suggestions that the adjuvant of aluminum in vaccines contributes to the occurrence of menstrual disturbances as an adverse event. Aluminum salts can enhance or prolong antigen-specific immune responses and suppress the concentrations of estrogen, progesterone, follicle stimulating hormone (FSH), and luteinizing hormone. They can also accumulate in the ovaries, causing structural damage, weight reduction, and decreased ovarian function. Furthermore, aluminum salts particularly influence HPV4, leading to amenorrhea, increased FSH levels, irregular menstruation, and premature menopause.[17] Anthrax vaccines showed a relatively small association with menstrual disorders, consistent with findings from previous studies.[18] However, considering the impact of vaccines on the immune system and menstrual periods there might be a slight association.[19] Vaccines associated with menstrual disorders showed the highest risk in the 0-11 and 45-64-year-old groups. According to previous studies, the 0-11 age group, being at the age of menarche, experiences more menstrual disorders due to the instability of menstrual cycles during this time.[20, 21] Additionally, the 45-64 age group, approaching menopause[22], is more prone to various menstrual disorders due to changes occurring during this life stage.[23] Therefore, these age groups appear to be more susceptible to vaccine-associated menstrual disorders, likely due to the natural vulnerability of their menstrual cycles around the times of menarche and menopause.
Vaccine-associated male sexual dysfunction had an association with the anthrax vaccine. However, there has been little prior research on this association with the exception of one article from 2002, importantly, this article was published over 20 years ago and consisted of a very s small sample size.[24] Therefore, it is challenging to analyze the mechanism by which the anthrax vaccine influences vaccine-associated male sexual dysfunction. Based on this paper, further research should be conducted. Our study suggested that vaccine-associated male sexual dysfunction has an association with COVID-19 mRNA vaccines. Numerous studies indicate that the SARS-CoV-2 virus, which causes COVID-19, influences male sexual dysfunction. Public single-cell RNA sequencing (scRNA-seq) datasets reveal a heightened expression of ACE2 and TMPRSS2 in various testicular cells, including spermatogonia, peritubular myoid cells, testis somatic cells, and spermatogonial stem cells. Therefore, the testis is considered highly susceptible to SARS-CoV-2 infection. Further studies using scRNA-seq data from male embryo primordial germ cells and normal testis cells underscored the ubiquity of ACE2 expression across all testicular cell types, including Sertoli cells. These results indicate that COVID-19 can harm both Sertoli and Leydig cells, suggesting that the SARS-CoV-2 virus may infiltrate the male reproductive system. Furthermore, analyses using histopathology, ultrastructural microscopy, and immunofluorescence of prostate tissue obtained during surgery for benign conditions from a male recovering from COVID-19 revealed viral particles in the prostate four months post-infection.[25] Therefore, it can be suggested that COVID-19 vaccines might also have an impact. However, there are no studies that suggest COVID-19 vaccines cause male sexual dysfunction, and some studies have argued that these vaccines do not cause male sexual dysfunction.[26] Furthermore, our study did not show a high association between COVID-19 vaccines and male sexual dysfunction, and given the limited prior research, further detailed studies are needed to explore this potential association.
Previous studies analyzed sexual disorders caused by vaccines separately or looked at differences in non-sexual diseases by sex. Additionally, these studies involved fewer participants compared to this research, resulting in relatively lower reliability and did not examine how sexual disorders vary by sex. Our research is the first to compare sexual disorders by sex, reinforcing the reliability of the results by involving a large number of people globally.
Similar to previous studies, the COVID-19 vaccines have shown a significant association with menstrual disorders. However, there is less research available on other vaccines, and many studies suggest that Anthrax vaccines do not have a significant association with menstrual disorders. Therefore, further research is needed. Additionally, there have not been many papers analyzing a wide range of vaccines simultaneously, indicating a need to study the associations of various vaccines with menstrual disorders together.
Most studies on menstrual disorders have excluded females younger than 18 years.[27] However, this research included those under 18 years, revealing significant associations were identified in the 0-11 age group for the first time. This highlights the necessity for research across all age groups. Additionally, most studies were conducted in high-income countries with extensive access to healthcare and resources for tracking and reporting menstrual disorders, limiting the generalizability of the results to other contexts.[28] The present study, however, analyzed data from nearly all countries, enabling more globally significant results.
In the case of male sexual dysfunction, there were few previous studies that had results contrary to this research.[26] Therefore, further investigation is needed to explore the mechanisms through which vaccines, as indicated by our findings, are linked to male sexual dysfunction.
Previous studies have explored sex differences in responses to various vaccines but did not address sexual disorders.[29] Therefore, this study analyzed sexual disorders arising from various vaccines based on sex. The results showed that different vaccines caused sexual disorders in males and females, and the mechanisms were also diverse. Thus, this suggests the necessity of considering these factors when administering vaccines.
Despite increasing reports of sexual disorders potentially caused by vaccines, research in this area has been lacking. Therefore, for a better understanding of menstrual disorders and male sexual dysfunction following vaccination, clinical follow-up is necessary.[7] These measures may increase confidence in vaccines, boost vaccination rates, and assist in achieving public health goals.
In addition, females around the onset or cessation of menstruation are particularly vulnerable to vaccine-associated menstrual disorders. Therefore, based on this study, there is a need for appropriate management and compensation for groups vulnerable to vaccines. Despite these adverse events, vaccination is essential to prevent more severe diseases in the future. Therefore, the Ministry of Health and Welfare should compare the benefits of vaccines with their adverse events, including menstrual disorders and male sexual dysfunction, to properly advise patients who need the vaccine and assist them in dealing with any adverse effects.
The present study looked at multiple vaccines simultaneously and included all age groups in the analysis, identifying significant results for vaccine-associated menstrual disorders and male sexual dysfunction for the first time. However, there are still some limitations.
First, because this study is cross-section by design,[30] it has the limitation that determining the cause of the results based on the analysis alone is difficult. However, because the analysis was conducted using previous studies on the relationship between vaccines and diseases as references, it is difficult to view the cause analysis as meaningless.
Second, there are limitations in the structure of the reporting system that analyzes vaccine-associated menstrual disorders. This study used passive reporting methods, which may lead to underrepresentation of vaccine-associated anaphylaxis cases and distort incidence rates. Therefore, the reported number of incidents likely lower than the actual occurrence.
Third, because menstrual disorders are a disease common among females, it is difficult to determine whether the menstrual disorders were caused by the vaccine. Therefore, further research is needed on the mechanisms underlying menstrual disorders associated with vaccines.
5. Conclusion
Analyzing the effects of different vaccinations on menstrual disorders and male sexual dysfunction, this study represents the first large-scale longitudinal research spanning. With a sample size of 7,903,767 individuals, it explores how various immunizations influence these health conditions. Although vaccines are associated with a rise in cases of menstrual disorders and male sexual dysfunction, the fatality rate linked to these adverse events remains minimal. Consequently, this significantly alleviates concerns regarding the safety of vaccinations. Our findings reveal that menstrual disorders were most significantly influenced by COVID-19 mRNA vaccines. Additionally, menstrual disorders also showed associations with papillomavirus vaccines, Ad5-vectored COVID-19 vaccines and anthrax vaccines. Among the vaccines identified, the risk was particularly higher in the 0-11 and 45-64-year-old age groups. In the case of male sexual dysfunction, the anthrax vaccine was most frequently associated, followed by the COVID-19 mRNA vaccines and Ad5-vectored COVID-19 vaccines. Based on these results, guidelines should be developed to tailor vaccination strategies, considering vaccines that can cause menstrual disorders and male sexual dysfunction, particularly in vulnerable age groups. The Ministry of Health and Welfare should also recommend vaccines based on their effectiveness and consideration of potential adverse events.







